
Jun 23, 2016
North America
Healthcare has always played a prominent role in presidential campaigns, and with good reasons. While the approaches and obstacles to reform can be addressed among multiple political parties, what remains undebatable is the fact that America’s healthcare system...
Read More

Jun 3, 2016
Asia
The China Food and Drug Administration (CFDA) has been changing how medical devices are regulated by expanding and updating their laws. They have released multiple draft documents which elaborate on the caliber of medical device clinical trials and devices that may be...
Read More

Jun 2, 2016
North America
The FDA is in the process of implementing the Unique Device Identification (UDI) System in a series of phases. Currently, all Class III and implantable, life-supporting and life-sustaining medical devices require UDIs. The next phase of the system will be implemented...
Read More
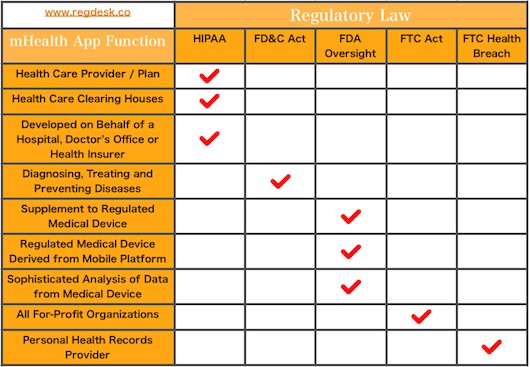
May 24, 2016
North America
Mobile health app The growing significance of mobile health apps result in a need to ensure its proper regulation. Practitioners, patients, researchers and medical industries are all affected by mHealth apps. The digital health field is regulated by the FDA under four...
Read More

May 11, 2016
Consultant Post
China has become the second largest medical device market in the world. Sustained economy, population growth and a burgeoning aging population makes China a lucrative market for foreign medical device and pharmaceutical manufacturers. Nevertheless, China is considered...
Read More
 North America
North America




