Feb 15, 2023
Uncategorized
Over the past few decades, there has been a significant evolution in medical devices in the healthcare industry. Medical devices are an important aspect of healthcare as they help diagnose, monitor, and treat a wide range of medical conditions. These devices include...
Read More

Feb 15, 2023
China
A Class III medical device is a device that is intended for use in the diagnosis, cure, treatment, or prevention of disease and is also intended to affect the structure or any function of the body. These devices are generally the most complex and pose the greatest...
Read More
Feb 15, 2023
Uncategorized
A class II medical device is a device that is intended to be used for a moderate-risk purpose. These devices may not pose the same level of risk as class III devices, but they still require some level of regulatory control to ensure their safety and effectiveness....
Read More
Feb 15, 2023
Uncategorized
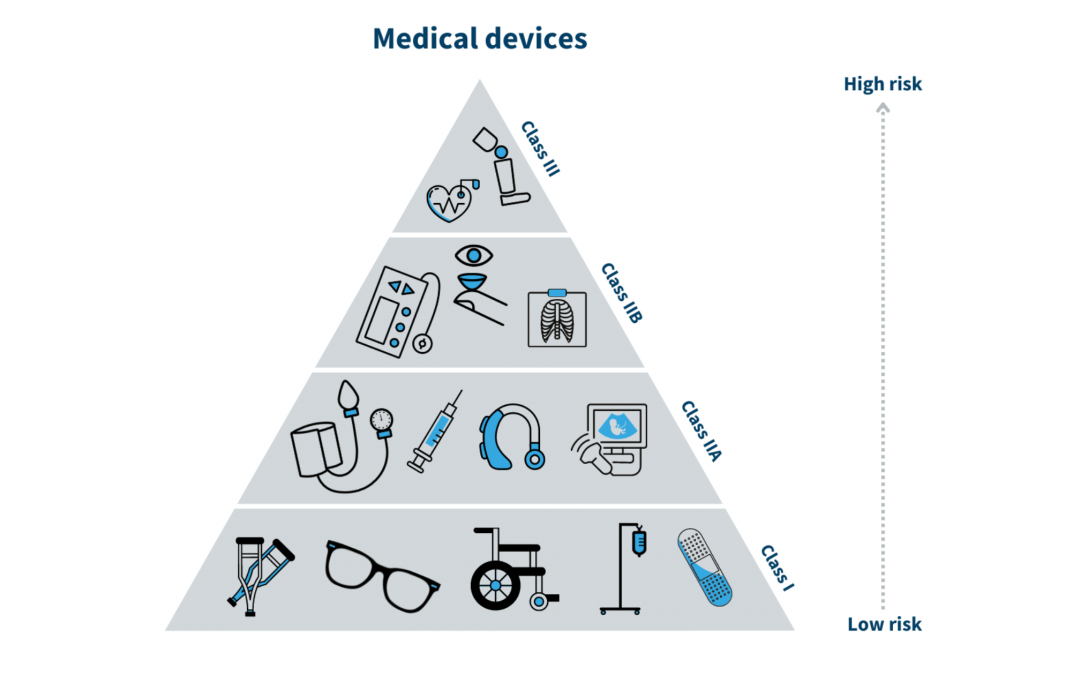
A medical device is a product that is used in the diagnosis, prevention, treatment, or management of a disease or medical condition. They are designed to assist healthcare professionals in their work and can range from simple instruments, such as a thermometer, to...
Read More

May 28, 2019
North America
The US FDA announced the launch of a pilot regulatory project related to the “Software as a Medical Device” (SaMD) in the course of amendments to the medical devices certification rules. 2019 Test Plan The development of technologies and regulatory needs are...
Read More
 Uncategorized
Uncategorized




