
Dec 19, 2023
Australia
The article highlights the aspects related to the inclusion of new products in the Australian Register of Therapeutic Goods (ARTG), a country’s register of healthcare products allowed for marketing and use in the country.
Read More

Dec 19, 2023
Australia
The Therapeutic Goods Administration (TGA), an Australian regulating authority in the sphere of healthcare products, has published a guidance document dedicated to the use of market authorization evidence from comparable overseas regulators/assessment bodies for medical devices (including in vitro diagnostic ones) for abridgement of TGA conformity assessments and as information required for applications for inclusion in the Australian Register of Therapeutic Goods (ARTG).
Read More
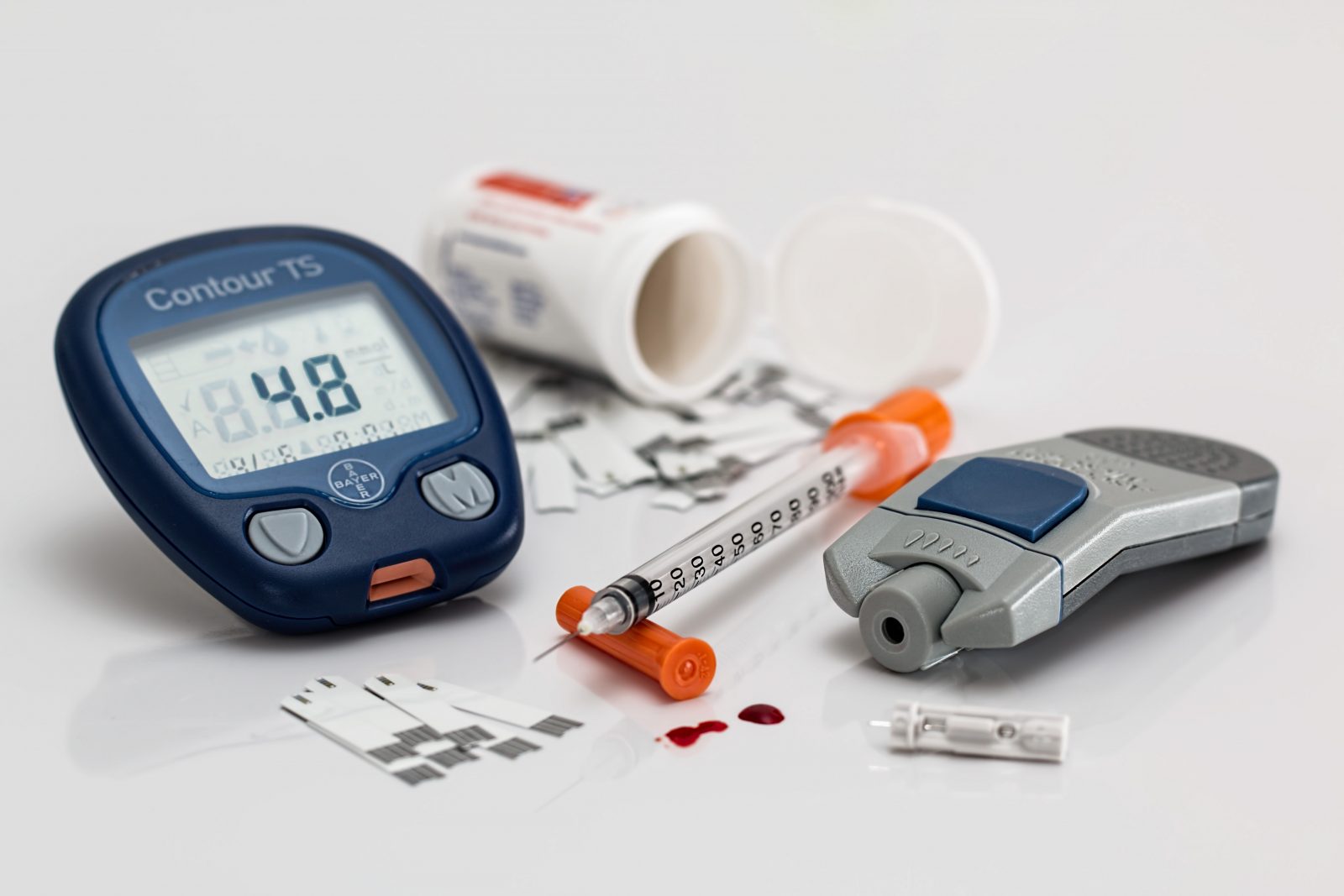
Sep 29, 2023
Asia
The article describes in detail the approach to be applied with respect to clinical evidence the parties responsible for medical devices are expected to provide when applying for marketing approval. Table of Contents The Health Sciences Authority (HSA), Singapore’s...
Read More

Jan 27, 2023
Canada
The new article highlights the key points related to comparator devices in the context of submitting clinical evidence when applying for marketing approval. Table of Contents Health Canada, a country’s regulatory agency in the sphere of healthcare products, has...
Read More

Jan 16, 2023
Canada
The new article highlights the aspects related to situations when clinical data is not needed, as well as to the changes to medical devices and amendments to the respective applications. Table of Contents Health Canada, a country’s regulating authority in the sphere...
Read More
 Australia
Australia




