
Sep 30, 2015
North America
Many manufacturers believe that selling their product or device without FDA approval is acceptable. If people buy it, but is this breach in ethics worth the potential consequences? If caught marketing an unapproved product, the FDA may issue a warning letter...
Read More

Sep 28, 2015
RegDesk News/Info
With emerging markets projected to account for 30% of global pharma market development, it’s important to note why having local consultants in those markets is vital. If not local, consultants are much more expensive. If a US consultant were hired for a project...
Read More

Sep 17, 2015
South America
Here are steps for proper device labeling in Brazil: The Brazilian Consumer protection code requires that product labeling should provide the consumer with accurate, concise and easily readable information. about the product’s quality, quantity, composition,...
Read More
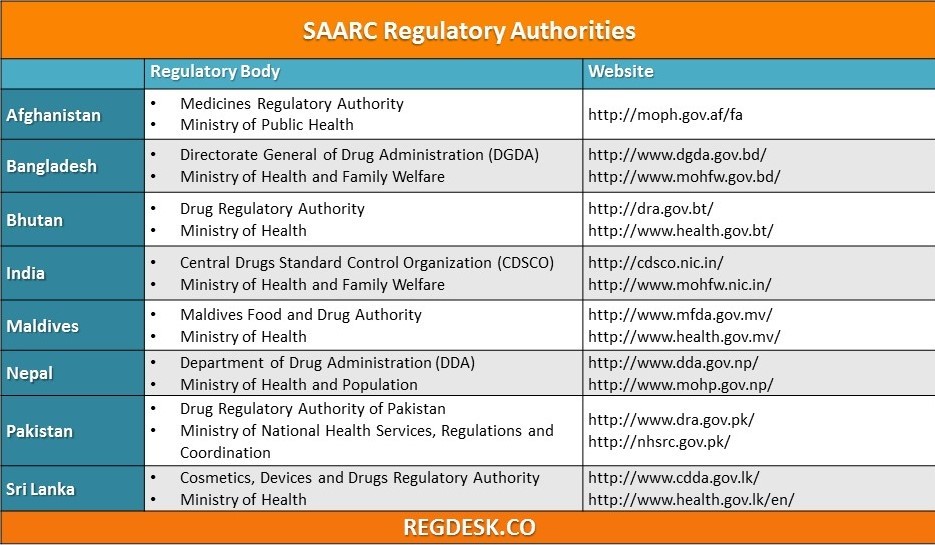
Aug 24, 2015
Asia
The chart above lists the pharmaceutical, medical and related regulatory authorities of countries who are members of the South Asian Association for Regional Cooperation along with links to their relevant websites. Also be sure to use our other reference charts...
Read More

Aug 13, 2015
Africa
Karen M, one of our Switzerland-based consultants, recently shared her insights into emerging markets of medical device with us. Here are Karen’s five tips to consider before sending your product to a new or developing market: Understand existing regulations...
Read More
 North America
North America




