Feb 15, 2023
FDA
A Class 1 medical device is a type of medical device that poses a minimal potential for harm to the user and is generally considered to be low-risk.Class 1 medical devices include a wide range of products, such as bandages, crutches, elastic bandages, and orthopedic...
Read More

Feb 15, 2023
China
A Class III medical device is a device that is intended for use in the diagnosis, cure, treatment, or prevention of disease and is also intended to affect the structure or any function of the body. These devices are generally the most complex and pose the greatest...
Read More
Feb 15, 2023
Uncategorized
A class II medical device is a device that is intended to be used for a moderate-risk purpose. These devices may not pose the same level of risk as class III devices, but they still require some level of regulatory control to ensure their safety and effectiveness....
Read More
Feb 15, 2023
Uncategorized
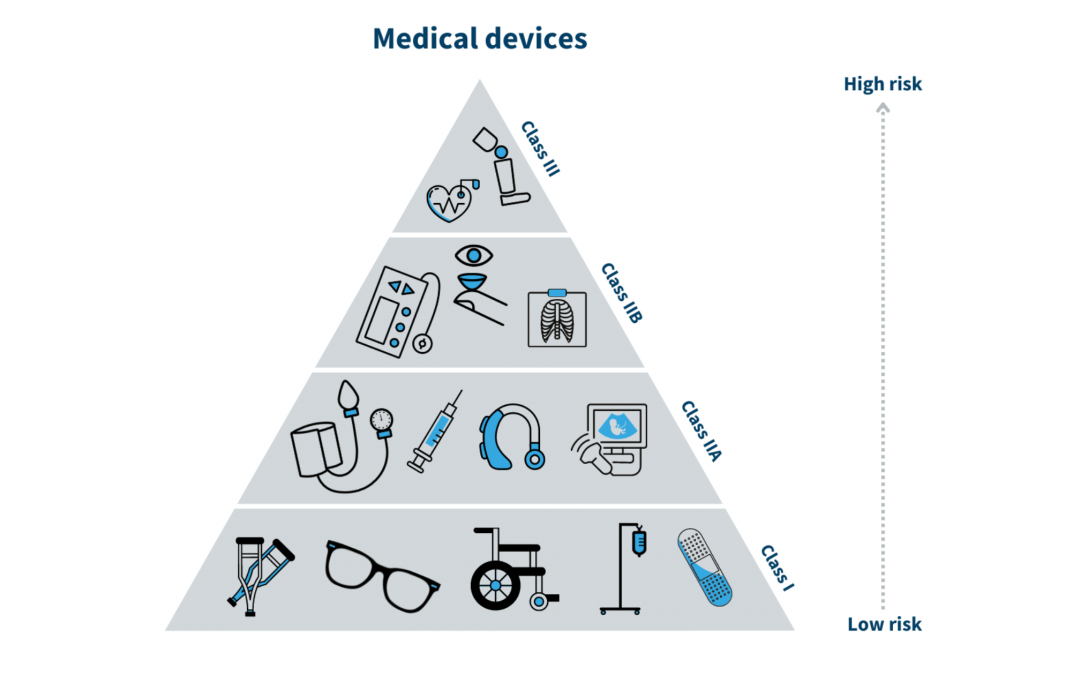
A medical device is a product that is used in the diagnosis, prevention, treatment, or management of a disease or medical condition. They are designed to assist healthcare professionals in their work and can range from simple instruments, such as a thermometer, to...
Read More

Feb 14, 2023
FDA
The new article highlights the questions related to the impact of the actions committed by the applicant on the FDA review clock. Table Of Contents: The Food and Drug Administration (FDA or the Agency), the US regulating authority in the sphere of medical devices, has...
Read More
 FDA
FDA




