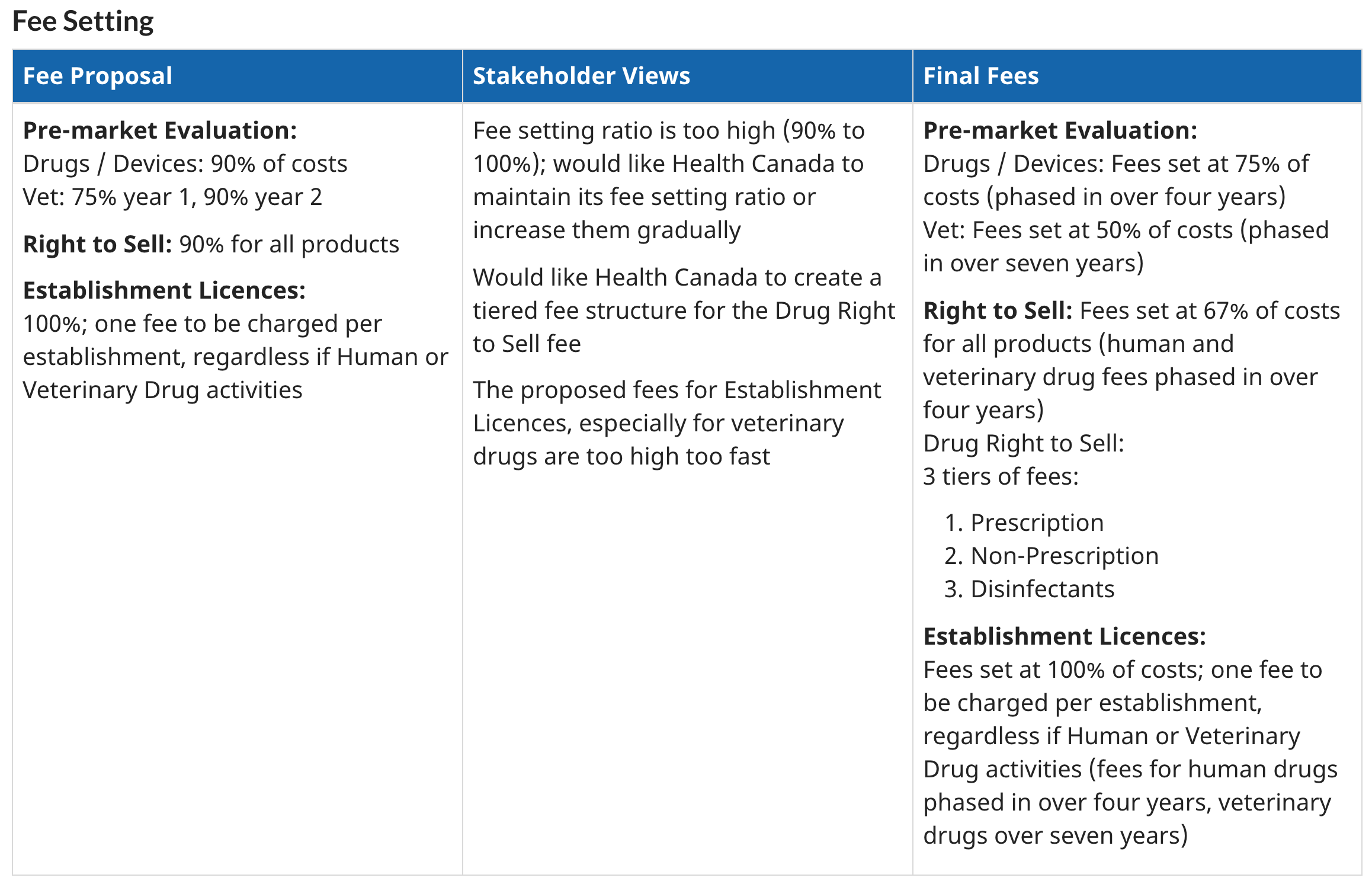
Overall, the regulatory cost increased by 43%.
Stakeholders of Health Canada stated that the fees should be increased in a more steady manner, rather than such abrupt change.

The basics of the regulation process in Canada (performed by Health Canada)
Submission / Application Evaluation (EVAL) Fees
Before a drug or medical device is authorized for sale in Canada, Health Canada reviews it to assess its safety, efficacy and quality.
Establishment Licensing (EL) fees
Health Canada inspects establishments to assess whether they comply with regulatory requirements to conduct regulated activities related to drugs and medical devices.
Right-to-Sell (RTS) fees
Health Canada monitors products on the Canadian market through post-market surveillance and compliance and enforcement activities.
Examination of application fees
Health Canada will increase fees regarding examination of application for each class of medical devices for the Fiscal Year 2020-2021.
Below is a detailed table of the fees.
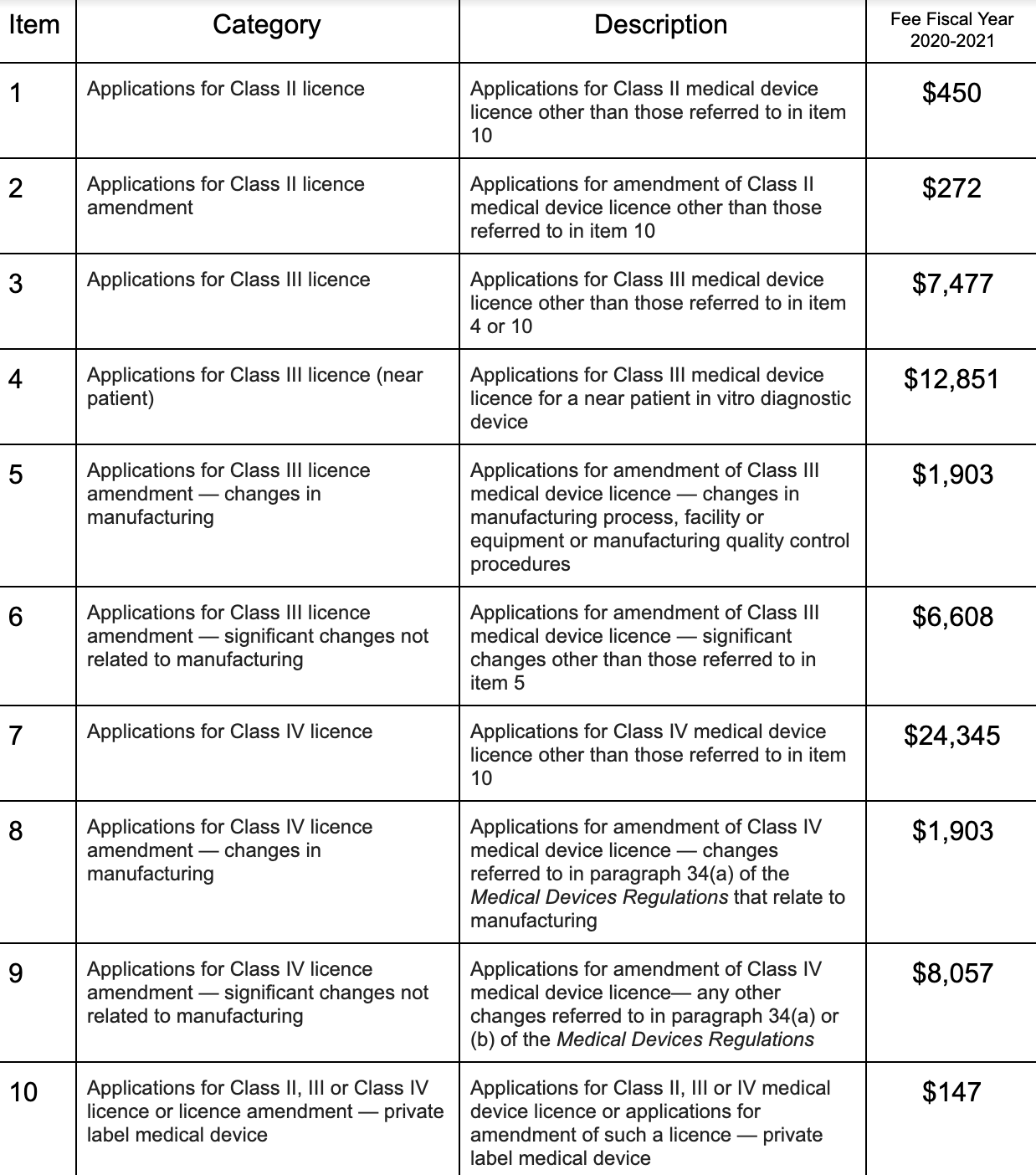
Retrieved from the Final Report published by Health Canada.
*Fees for examination of application for an establishment license – Medical Devices
Medical Devices Establishment License
– Application for new license and annual review of license
$4590
(120 calendar days to issue decision)
*Fees for the Right-To-Sell (RTS) licensed class II, III, or IV devices
Right-To-Sell medical device
– The annual fee to right to sell medical devices (class II, III, or IV).
$381
(20 days to update Medical Device Licence Listing database following receipt of a complete Annual Notification Package.)
Other finalized significant changes in fees:
Small Business Strategy
–Applying the Treasury Board Secretariat’s small business definition (and the Competition Act’s definition of affiliates):
-First Pre-market submission free regardless of fee amount
– 50% mitigation for all subsequent Pre-market Evaluation fees
– 25% mitigation for all Right to Sell fees
– 25% mitigation for all Establishment License fees
Costing
Costing document detailing methodology
Tiered Drug Right to Sell fee
Annual Fee Adjustment
–Annual fee adjustment tied to CPI of previous year, published in the fall with implementation April 1st
Small Business Strategy
Applying the Treasury Board Secretariat’s small business definition (and the Competition Act’s definition of affiliates):
– First Pre-market submission free regardless of fee amount
– 50% mitigation for all subsequent Pre-market Evaluation fees
– 25% mitigation for all Right to Sell fees
– 25% mitigation for all Establishment Licence fees
Mitigation Measures
Waive first Pre-market drug submission fee for a drug on the List of Drugs for an Urgent Public Health Need, as per the Access to Drugs in Exceptional Circumstances Regulations
Elimination of fee deferrals and mitigation based on product sales
All fees waived for publicly funded health care institutions
Drug Establishment Licence fees pro-rated quarterly for a new application
Performance Standards and Reporting
All existing standards will remain unchanged, except for:
– Human Drug Evaluation fee categories Labelling Only (120 days) and DINA Labelling Standard (60 days)
– Human Drug and Veterinary Drug Right to Sell fees (20 days)
– Disinfectant – Labelling Only (90 days)
All new fee categories have a performance standard
Provide quarterly and annual performance reports upon request
Annual meeting with industry to discuss performance, efficiencies and other areas of interest
Penalty Provision
–An individual submission or application that exceeds the performance standard will receive a rebate of 25%
Penalties will not apply to joint and parallel review submissions or medical device combination applications.
Consultations on the ‘Pause the Clock’ provisions concluded and the final guidance will be implemented April 1, 2020
Timing of Payment
-Full premarket fee collected upfront
Health Canada’s core principle behind raising the fees are as follows:
Health Canada’s main reasoning behind revising these fees are as below:
Be Reasonable and Fair: recognize that industry needs to pay its fair share and reduce the burden on taxpayers, fees have been set reasonably and are being phased-in
Minimize Impact on Small Business: fees should not deter small businesses from doing business in Canada
Apply Appropriate Mitigation: fees should be reduced or exempted in explicit circumstances to support the health care system
Make Fee Increases Gradual and Predictable: revised fees will be phased-in over multiple years
Ensure Accountability: remain transparent and accountable to stakeholders through annual reporting and annual engagement is key to developing an agile and responsive fee strategy
Health Canada will make these changes by April 2020 and stakeholders can provide feedback or any opinions about what they would think is crucial or necessary in making these changes.

