Centralized Tracking and Global Reporting
Say goodbye to scattered data in spreadsheets and missed deadlines. Our platform centralizes your medical device registration information, giving you real-time visibility into global approvals and renewals while enhancing cross-functional team alignment. With automated notifications, you’ll stay ahead of renewal deadlines, reduce penalties, and streamline your entire registration process. Boost efficiency and manage global submissions seamlessly.
Get a Demo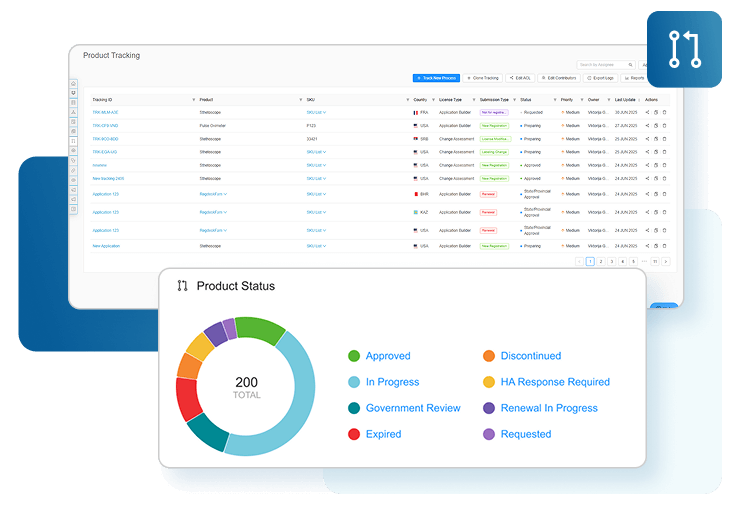
Always-On Compliance Management
Take charge of compliance with proactive management. Automatically track product timelines and receive reminders for upcoming renewals and health authority responses. With real-time updates, medical device manufacturers can effortlessly stay compliant across global markets while ensuring uninterrupted patient access to your company’s innovative product.
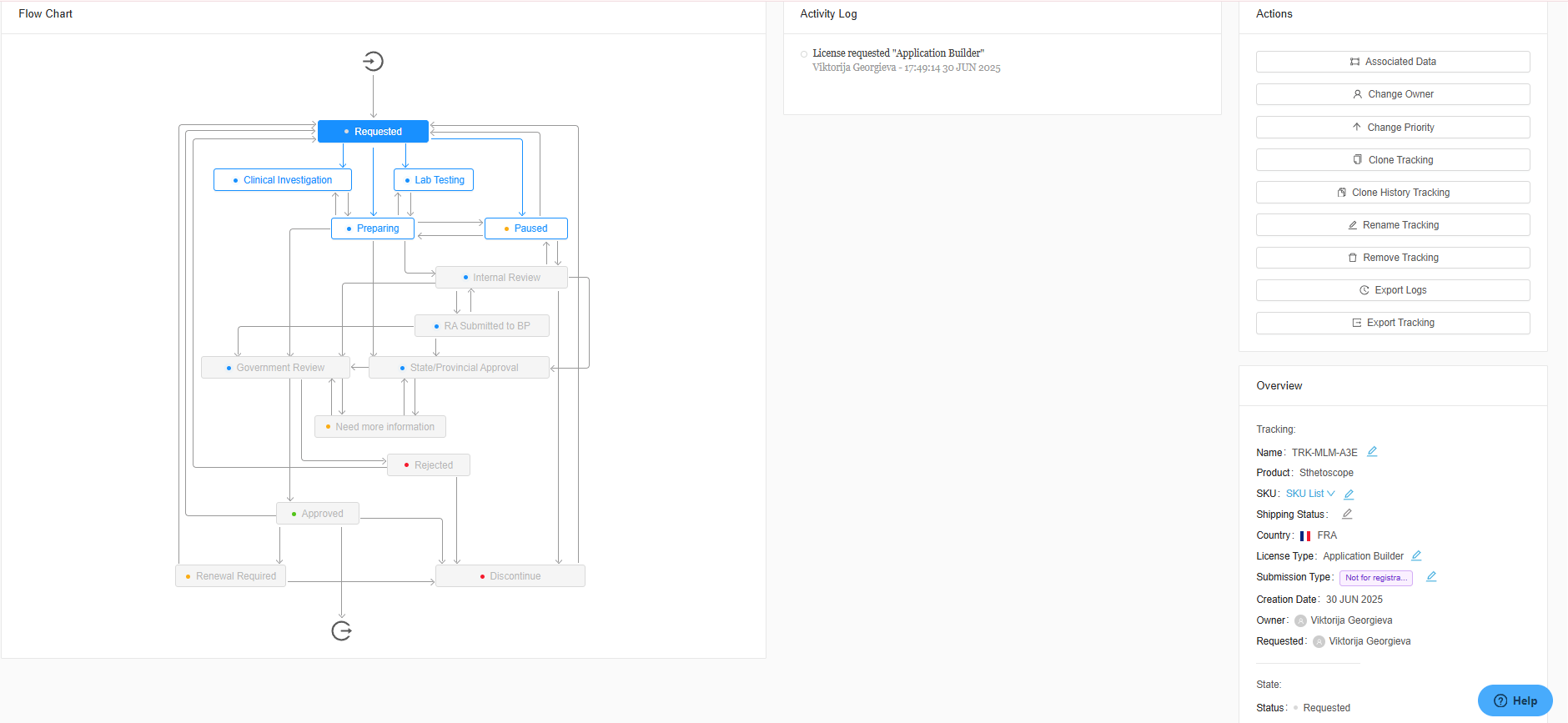
Smarter Decisions with Real-Time Analytics
RegDesk’s reporting and analytics tools offer customizable reports and real-time insights into key metrics like submission times, costs, and statuses. With automated renewal notifications and performance tracking, teams can track KPI’s, optimize workflows, and stay ahead of deadlines to ensure compliance. These powerful tools help manufacturers make data-driven decisions, streamline operations, and minimize penalties.

Seamless Regulatory Management
RegDesk offers an intuitive interface with centralized registration management, easy data access, and customizable reporting. Ensure compliance and optimize workflows with real-time insights, automated renewal notifications, and KPI tracking. With advanced user management and robust analytics, RegDesk streamlines regulatory workflows, improving efficiency and reducing the risk of non compliance.
“RegDesk’s built-in reporting and statistics in their Tracking module allows our regulatory leader to easily present the KPIs to the organization. We are also able to keep track of the incoming international requests which allowed for our company to enhance its approach on global expansion.”
Put an end to costly delays!
Get Started with RegDesk and experience enhanced visibility, proactive compliance,
streamlined workflows, and powerful analytics for your team.
Frequently Asked Questions
What is regulatory compliance software?
Regulatory compliance software helps organizations comply with legal, security and other regulatory requirements. The software automates and streamlines processes to make sure internal policies are followed and external requirements are met. Common features of this type of software include tools for risk assessment, compliance tracking, audit management and reporting capabilities. In industries like healthcare, finance or manufacturing, where there are strict regulations, regulatory compliance software is a must. In order to reduce non-compliance penalties, ensure accurate reporting, and increase operational efficiency companies can deploy a centralized system so they stay updated with evolving regulations and requirements.
What is regulatory compliance for medical devices?
Regulatory compliance for medical devices refers to upholding laws and regulations designed by the governing bodies in relation to design, testing, production and marketing of medical devices. Governing bodies, for example, Health Canada or US FDA, want to ensure medical device safety and efficacy for patients so they set standards medical device companies must follow. Companies must adhere to strict compliance requirements like pre-market approval, clinical testing, post-market surveillance, and adverse event reporting, to legally sell their products in various countries. This process protects both manufacturers and patients while ensuring products are safe and reliable.
Why is a regulatory strategy in medical devices important?
A regulatory strategy for medical devices is important because it provides medical device manufacturers with a plan to navigate the complex regulatory landscape. With diverse requirements across different markets, it is imperative companies follow the most efficient path to market approval and compliance. Having a strategic plan in place allows companies to manage compliance risks, reduce costs and become more efficient in their regulatory submission and approval process. By ensuring regulatory obligations are met, companies can avoid costly delays and non-compliance issues which can provide a competitive advantage and build trust with stakeholders and patients alike.
What is the regulatory path for medical devices?
The regulatory pathway for medical devices varies across regions. Generally, the first step in this complex process is to determine the risk classification of the medical device because this affects the regulatory requirements and submission pathway. When you know the device classification and target markets, it is important to understand the regulatory requirements you must follow. There will be preclinical testing, risk assessment, clinical evaluation and evidence gathering that you will need to submit your application. Once you have all of your documentation, you must apply and obtain market approval. After approval, it is imperative that you follow post-market surveillance guidelines and report any adverse events which is an ongoing process for compliance.





