
Jun 7, 2017
Asia
China Food and Drug Administration (CFDA) recently proposed policies to streamline and accelerate drug and medical device approvals as part of its regulatory reform. Below are highlights of some noteworthy changes that impact new drug and device approval...
Read More
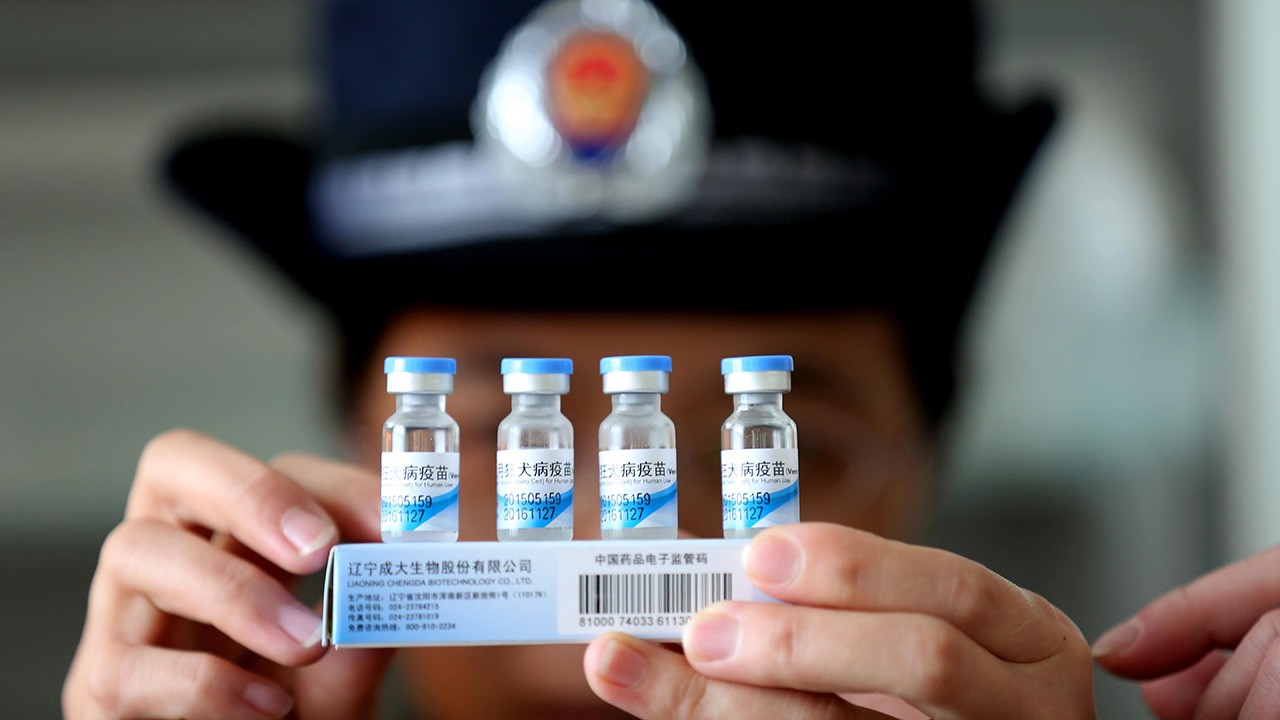
Jul 14, 2016
Asia
In April, China was hit by a vaccine scandal that has since resulted in a shortage of supply. Thousands of vaccine doses for children and adults were found to be improperly stored or expired. The vaccines were illegally sold for millions of dollars on the black...
Read More

Jun 3, 2016
Asia
The China Food and Drug Administration (CFDA) has been changing how medical devices are regulated by expanding and updating their laws. They have released multiple draft documents which elaborate on the caliber of medical device clinical trials and devices that may be...
Read More

May 11, 2016
Consultant Post
China has become the second largest medical device market in the world. Sustained economy, population growth and a burgeoning aging population makes China a lucrative market for foreign medical device and pharmaceutical manufacturers. Nevertheless, China is considered...
Read More

Jan 19, 2016
Asia
The Chinese Food and Drug Administration (CFDA) has issued a new “Medical Equipment Generic Naming Guide,” which will be implemented on April 1, 2016. This is the first regulatory document for ‘naming’ generic medical devices. The CFDA believes...
Read More
 Asia
Asia




